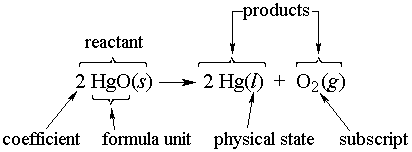
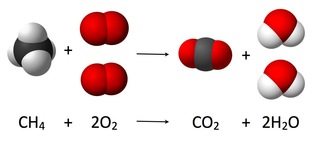
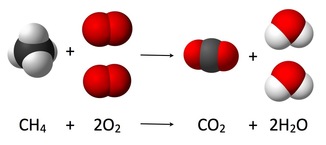
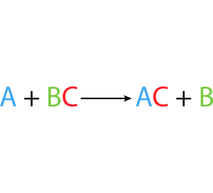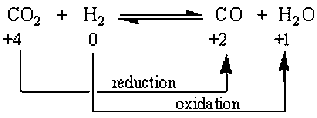Endothermic Reactions
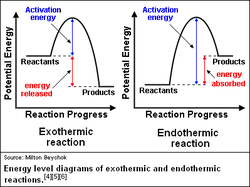
An endothermic reaction's products have more energy than the reactants, therefore energy is written on the product side. Energy or heat is absorbed and the reaction will feel cold.
Exothermic Reactions
An exothermic reaction's products have less energy than the reactants, which means energy is written on the reactant side. Heat is given off and the reaction feels hot.
An endothermic reaction's products have more energy than the reactants, therefore energy is written on the product side. Energy or heat is absorbed and the reaction will feel cold.
Exothermic Reactions
An exothermic reaction's products have less energy than the reactants, which means energy is written on the reactant side. Heat is given off and the reaction feels hot.
Law of Conservation of Mass and Energy
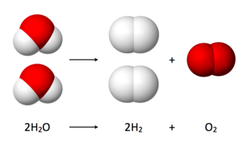
Law of conservation of mass states that mass cannot be created nor destroyed, but it can be transferred. The number of atoms does not change. The total number one type of atom on the reactant side must equal the amount on the product side.
Law of conservation of energy states that energy cannot be created nor destroyed, but it can be transferred. Same as the conservation of mass, the total amount of energy on the reactant side must equal that on the product side. Energy is found in the bonds between atoms and is either released or absorbed during the reaction.
These laws are important because they tell us that both sides of a reaction have to be equal.
Types of Chemical Reactions
There are six types of chemical reactions: combustion, synthesis, decomposition, single displacement, double displacement, and oxidation.
Combustion
When oxygen combines with another compound to form water and carbon dioxide, a combustion reaction occurs. These reactions are exothermic, meaning they produce heat.
Law of conservation of mass states that mass cannot be created nor destroyed, but it can be transferred. The number of atoms does not change. The total number one type of atom on the reactant side must equal the amount on the product side.
Law of conservation of energy states that energy cannot be created nor destroyed, but it can be transferred. Same as the conservation of mass, the total amount of energy on the reactant side must equal that on the product side. Energy is found in the bonds between atoms and is either released or absorbed during the reaction.
These laws are important because they tell us that both sides of a reaction have to be equal.
Types of Chemical Reactions
There are six types of chemical reactions: combustion, synthesis, decomposition, single displacement, double displacement, and oxidation.
Combustion
When oxygen combines with another compound to form water and carbon dioxide, a combustion reaction occurs. These reactions are exothermic, meaning they produce heat.
Synthesis
Two or more simple compounds combine, meaning a synthesis reaction.
Example:
Two or more simple compounds combine, meaning a synthesis reaction.
Example:
Decomposition
Complex compounds can break apart to form simpler compounds, called decomposition. Decomposition is the opposite of synthesis.
Complex compounds can break apart to form simpler compounds, called decomposition. Decomposition is the opposite of synthesis.
Single Displacement
One element may switch places another element in a compound- this is called single displacement.
One element may switch places another element in a compound- this is called single displacement.
Double Displacement
When cations and anions switch places in two different molecules and form two new compounds, it is called double displacement.
When cations and anions switch places in two different molecules and form two new compounds, it is called double displacement.
Oxidation
When oxygen combines with other compounds and water is produced, it is called oxidation.
When oxygen combines with other compounds and water is produced, it is called oxidation.
Factors that increase reactions
Chemical reactions may be sped up by increasing the number of collisions. The number of collisions may be increased by increasing surface area, temperature, stirring, and using a catalyst.
Chemical reactions may be sped up by increasing the number of collisions. The number of collisions may be increased by increasing surface area, temperature, stirring, and using a catalyst.
- Surface area- by increasing surface area, the particles are crushed and have more area for the chemicals to react.
- Temperature- increasing the temperature, increases the motion.
- Stirring- stirring increases motion.
- Catalysts- catalysts increase the reaction without affecting the reaction.
Equilibrium
When the reactants combine to form products at the same rate that the products break down to form products, it has reached equilibrium. Changing the pressure, temperature, concentration, of the reactants or products shifts the equilibrium and the reaction is no longer equal.
When the reactants combine to form products at the same rate that the products break down to form products, it has reached equilibrium. Changing the pressure, temperature, concentration, of the reactants or products shifts the equilibrium and the reaction is no longer equal.