Chapter 4: Atoms & The Periodic Table
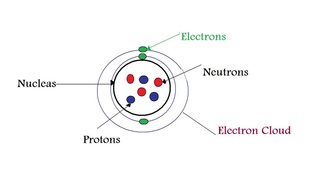
Atoms
What Changes Happen In An Atom That Makes Them Stable:
- Atoms Bond To Form Compounds Or Molecules.
Electronegativity Trend (What It Means):
- From Top To Bottom Down A Group, Electronegativity Decreases. This Is Because Atomic Number Increases Down A Group, And Thus There Is An Increased Distance Between The Valence Electrons And Nucleus, Or A Greater Atomic Radius.
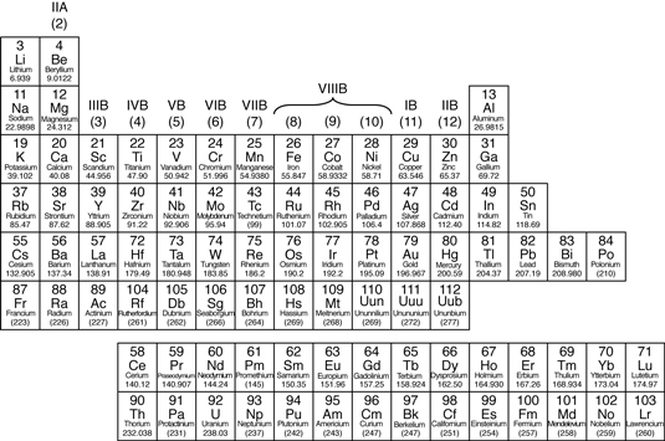
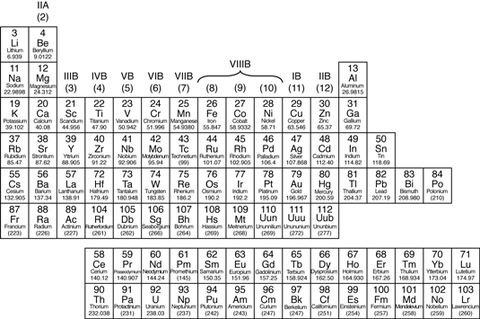
The Periodic Table
|
Periodic Table: A table in which the chemical elements are arranged in order of increasing atomic number. Elements with similar properties are arranged in the same column (called a group), and elements with the same number of electron shells are arranged in the same row (called a period).
Group: in chemistry, a set of chemical elements in the same vertical column of the periodic table. The elements in a group have similarities in the electronic configuration of their atoms, and thus they exhibit somewhat related physical and chemical properties. Arranged by increasing elements. |
|
Metals
In the periodic table, you can see a stair-stepped line starting at Boron (B), atomic number 5, and going all the way down to Polonium (Po), atomic number 84. Except for Germanium (Ge) and Antimony (Sb), all the elements to the left of that line can be classified as metals.
These metals have properties that you normally associate with the metals you encounter in everyday life:
· They are solid (with the exception of mercury, Hg, a liquid).
· They are shiny, good conductors of electricity and heat.
· They are ductile (they can be drawn into thin wires).
· They are malleable (they can be easily hammered into very thin sheets).
All these metals tend to lose electrons easily. The following figure shows the metals.
These metals have properties that you normally associate with the metals you encounter in everyday life:
· They are solid (with the exception of mercury, Hg, a liquid).
· They are shiny, good conductors of electricity and heat.
· They are ductile (they can be drawn into thin wires).
· They are malleable (they can be easily hammered into very thin sheets).
All these metals tend to lose electrons easily. The following figure shows the metals.
Nonmetals
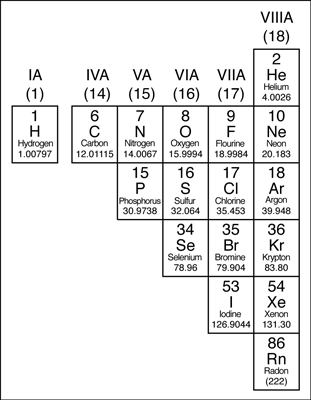
Except for the elements that border the stair-stepped line, the elements to the right of the line are classified as nonmetals (along with hydrogen). Nonmetals have properties opposite those of the metals.
The nonmetals are brittle, not malleable or ductile, poor conductors of both heat and electricity, and tend to gain electrons in chemical reactions. Some nonmetals are liquids. These elements are shown in the following figure.
The nonmetals are brittle, not malleable or ductile, poor conductors of both heat and electricity, and tend to gain electrons in chemical reactions. Some nonmetals are liquids. These elements are shown in the following figure.
Metalloids
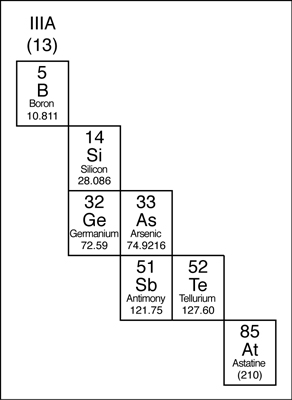
The elements that border the stair-stepped line are classified as metalloids. The metalloids, or semimetals, have properties that are somewhat of a cross between metals and nonmetals.
Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry. The metalloids are shown in the following illustration.
Metalloids tend to be economically important because of their unique conductivity properties (they only partially conduct electricity), which make them valuable in the semiconductor and computer chip industry. The metalloids are shown in the following illustration.
Source: http://www.chemicool.com/