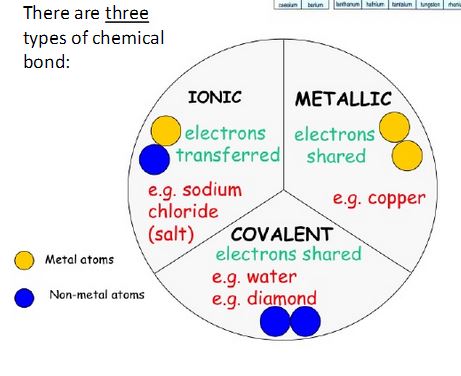
There are three types of bonds which are covalent, metallic, and ionic. They have different force attractions. There electrons are shared between different atoms and different metals.
Covalent Bonds- Force of attraction is between positive nucleus of one atom and negative electron cloud of another atom. Electrons are shared between a few atoms. They are shared between nonmetals and
nonmetals.
Metallic Bonds- Force of attraction is between positive nucleus of one atom and negative electron cloud of
another atom. Electrons are shared between all atoms. They are shared between metals and metals.
Ionic Bonds- Force of attraction is between opposite charges. Electrons are donated or accepted. They are shared between metals and nonmetals.
http://facstaff.gpc.edu/~pgore/PhysicalScience/Naming-chemical-compounds.html
Covalent Bonds- Force of attraction is between positive nucleus of one atom and negative electron cloud of another atom. Electrons are shared between a few atoms. They are shared between nonmetals and
nonmetals.
Metallic Bonds- Force of attraction is between positive nucleus of one atom and negative electron cloud of
another atom. Electrons are shared between all atoms. They are shared between metals and metals.
Ionic Bonds- Force of attraction is between opposite charges. Electrons are donated or accepted. They are shared between metals and nonmetals.
http://facstaff.gpc.edu/~pgore/PhysicalScience/Naming-chemical-compounds.html
